Can we Create New Life?
- Atharv Sharma
- Feb 28
- 6 min read
Author: Atharv Sharma
In our journey to understand what constitutes life, we began with the recognition that life is not a static state, but a process that is continuous, regulated, and governed by fundamental physical laws. At the core of this process is the Second Law of Thermodynamics, which states that the entropy of the universe tends to increase. Living systems, in contrast, maintain low internal entropy through highly ordered structures and regulated biochemical reactions. To sustain this order, organisms consume external energy, such as light, chemical bonds, and nutrients, and in doing so, contribute to an overall increase in universal entropy. This thermodynamic law underpins not only metabolism, but also growth, reproduction, and evolution. We explored how life exhibits organic chemistry, which are primarily carbon-based, due to carbon’s molecular versatility and its chemical properties, and how living entities' organization is essential for function. Without this vital order of life, even a chemically intact system ceases to be alive. We examined the pre-programmed nature of organisms through their genetic instructions, their ability to maintain homeostasis, interact with environments, and adapt through mutations in their genomes. Reproduction and evolution emerged as key traits for the continuity and diversification of life. This framework grounded our understanding as we progressed toward evaluating whether new life can be artificially created.

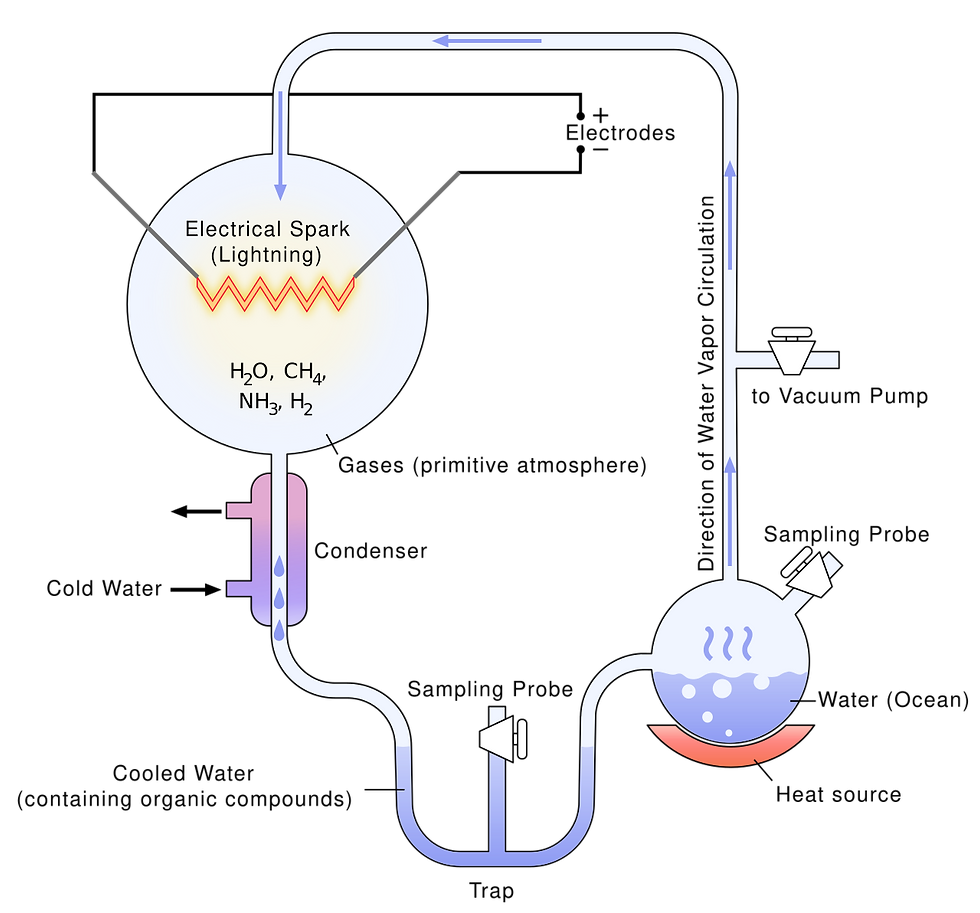
Following our thermodynamic framework for life, we turned to the question of abiogenesis: could life arise from non-life under natural conditions? We examined decades of experiments that aim to simulate and model a prebiotic Earth and reconstruct the molecular steps that might have given rise to the first living systems. Prebiotic chemistry has made demonstrable progress. The 1952 Miller–Urey experiment, performed by Stanley Miller and Harold Urey, showed that amino acids (the monomers of proteins) can form when simple gases like methane, ammonia, and hydrogen are exposed to electrical discharges, which aimed to simulate lightning in the early earth atmosphere. The experiments that under varied atmospheric conditions have produced additional building blocks such as nucleotides, lipids, and simple sugars. These results support the notion that abiotic synthesis of life’s molecular components is chemically attainable.
However, synthesising components does not mean we can create life. The RNA world hypothesis, which posits that early life relied on RNA both to store genetic information and to catalyse its own replication, represents a significant conceptual advance. Laboratory experiments have produced ribozymes, which is an RNA molecules with enzymatic activity, that can catalyse ligation and, in some cases, template-directed RNA polymerisation. Though some ribozymes can extend complementary strands, no current system achieves sustained, error-tolerant replication cycles with open-ended evolution. Thus, while the RNA-first model is chemically plausible, it has not yet bridged the gap from molecular function to autonomous inheritance and selection.
To further test the plausibility of abiogenesis, researchers have constructed model protocells, that are self-assembling lipid vesicles that encapsulate RNA and other macromolecules. Fatty acid vesicles can grow by incorporating additional lipids from their environment and can divide under mechanical forces, mimicking primitive cell division. Some of these vesicles can maintain internal pH, support encapsulated reactions, and even accommodate ribozyme-mediated RNA replication under specific conditions. Nonetheless, such systems remain far from autonomous: they require carefully controlled laboratory inputs and do not exhibit full thermodynamic coupling between replication, metabolism, and compartmentalization. Thus, while we can generate life-like behaviours and systems that imitate early biogenesis, a self sustaining, evolving cell built entirely from abiotic inputs has not yet been achieved.

We then considered the possibility of constructing life from wholly inorganic systems. If life is ultimately about thermodynamic structure, organisation, and information flow, then perhaps it is not bound to carbon chemistry. In this context, we studied the work of researchers who developed inorganic chemical cells, iCHELLs, assembled from metaloxide clusters known as polyoxometalates. These systems exhibit membrane-bound compartmentalisation, selective ion exchange, and redox-coupled internal reactions. iCHELLs can form nested vesicles, generate electrochemical gradients, and perform simple energy storage, which are foundational properties of biological membranes. However, no iCHELL system has demonstrated autonomous replication, mutation, or Darwinian evolution. Thus, they remain important experimental models for artificial life, but not yet life itself. Their significance lies in the fact that they expand the conceivable chemistry of life beyond carbon and water, opening theoretical windows for life on other planets or in synthetic ecosystems.
Parallel to these biochemical experiments, we explored the frontiers of synthetic bioengineering. One of the most compelling examples is the creation of Xenobots, precisely defined as living, motile assemblies built entirely from differentiated frog cells, shaped and arranged using algorithmic design. Unlike protocells or iCHELLs, Xenobots are not based on abiotic synthesis but rather reprogramming biological materials. Yet their design and function are wholly unique as they do not exist in nature and do not reproduce through conventional biological means. What we found remarkable was their capacity for collective motion, environmental responsiveness, and in certain cases, self-replication by kinematic aggregation of surrounding cells into daughter forms. This replication does not rely on DNA or cellular division, but on emergent mechanical assembly and cell cohesion behaviours.

Collectively, our review led us to a precise and cautiously optimistic conclusion. No current experiment nor our hypothesis have demonstrated the spontaneous creation of a fully autonomous, evolving cell from purely abiotic starting materials. Yet, many partial systems—protocells, ribozymes, inorganic vesicles, minimal genomes, and programmable cell aggregates—have shown distinct life-like properties: replication, metabolism, movement, and information storage. Each system satisfies some, but not all, of life’s requirements. Whether a laboratory construct qualifies as “life” depends not only on its properties but on the philosophical and operational definitions applied.
In our view, we have not yet created life. But we are not fundamentally constrained by known laws of physics or chemistry. Rather, we are limited by technological capacity, systems integration, and incomplete understanding of life’s minimal thresholds. As we improve microfluidics, automated evolution platforms, and computational design, we anticipate that integrated, fully abiotic life will become more attainable. Life has not been created yet—but every building block we assemble brings that threshold closer within reach.
Glossary
Entropy: A measure of disorder or randomness in a system; the Second Law of Thermodynamics states that entropy naturally increases over time, which living systems counteract by using energy to maintain internal order.
Abiogenesis: The scientific hypothesis that life originated from nonliving chemical compounds under early Earth conditions without supernatural intervention.
Primordial soup: A conceptual model describing Earth’s early oceans as chemically rich environments where organic molecules accumulated and interacted.
Miller–Urey experiment: A landmark 1952 experiment demonstrating that organic molecules, such as amino acids, can form spontaneously from simple gases when exposed to energy sources like lightning.
RNA world hypothesis: The proposal that early life relied on RNA molecules for both storing genetic information and catalysing chemical reactions, predating DNA and proteins.
Ribozyme – A catalytic RNA molecule capable of facilitating biochemical reactions, supporting the idea that RNA could have driven early self-replication.
Template-directed polymerisation – The process by which a nucleic acid strand guides the formation of a complementary strand, foundational for genetic inheritance.
Protocell – A primitive, cell-like structure usually composed of lipid membranes enclosing biochemical reactions, thought to be precursors to living cells.
Lipid vesicle – A spherical membrane structure made from fatty acids or phospholipids capable of encapsulating molecules, modelling early cellular compartments.
Compartmentalisation – The separation of biochemical processes into distinct spaces, enabling organisation, stability, and metabolic coordination in living cells.
iCHELL (Inorganic Chemical Cell) – An artificial, non-carbon-based compartment assembled from metal-oxide clusters (polyoxometalates) that mimics some features of biological cells (ion exchange, membranes, gradients).
Polyoxometalates (POMs) – Inorganic molecular clusters used to construct iCHELL systems, demonstrating life-like structure without carbon chemistry.
Synthetic bioengineering – The design and construction of biological systems and organisms with novel properties, often using computational tools and cellular reprogramming.
Xenobot – A biologically constructed organism made from frog cells, engineered via computational models; capable of autonomous motion and, in some cases, kinematic self-replication.
Comments